¶ Evidence Based Analysis Of Frontier Longevity Interventions
¶ Summary
Frontier longevity interventions are emerging strategies aimed at extending lifespan and improving healthspan. These interventions include caloric restriction, dietary modifications, exercise protocols, rapamycin, metformin, senolytics, and NAD+ boosters. Recent studies suggest that these interventions may have significant effects on aging and longevity, although more research is needed to fully understand their efficacy and safety.
¶ Evidence Summary: Human Outcomes
The following table summarizes the quality of evidence for key longevity interventions, focusing specifically on human clinical outcomes. Unlike animal studies, human data for lifespan extension is generally unavailable; therefore, proxies such as biological aging clocks, metabolic health, and functional capacity are the primary endpoints.
| Intervention | Human Clinical Outcomes (Key Trials) | Evidence Quality (GRADE) | Safety Profile |
|---|---|---|---|
| Caloric Restriction | Slowed Aging Pace: 2 years of ~12% CR slowed biological aging (DunedinPACE) by ~2-3% and improved cardiometabolic risk scores (CALERIE-2 Trial) [1][2]. | High (RCT) | Safe: Monitored CR is safe; risks include loss of bone density and libido if excessive. |
| Exercise Protocols | Mortality Reduction: High cardiorespiratory fitness (VO2 max) associated with >40% lower all-cause mortality vs. low fitness [3]. Combination: Aerobic + Resistance training provides superior mortality reduction compared to either alone [4]. |
High (Meta-analyses of Cohorts & RCTs) | High Safety: Injury risk exists but benefits outweigh risks. |
| Dietary Interventions (Mediterranean) | Disease Prevention: Significant reduction in cardiovascular events (PREDIMED) and cognitive decline; associated with lower all-cause mortality [5]. | High (RCT & Meta-analyses) | High Safety: Gold standard for healthy eating. |
| Rapamycin | Immune Function: Improved response to influenza vaccine (20% increased antibody titers) and reduced infections in elderly adults (Mannick et al.) [6][7]. Lifespan: Insufficient human data. |
Moderate (Phase 2 RCTs for Immune function) | Moderate Risk: Mouth sores, glucose elevation, potential immune suppression at high doses. |
| Metformin | Disease Risk: Reduced cancer/CVD/mortality in diabetic populations (Observational) [8]. Exercise Blunting: Blunted mitochondrial adaptations and VO2max gains in healthy older adults (Konopka et al.) [9]. Lifespan: Insufficient human data (TAME trial pending). |
Low (Observational for longevity) High (RCT for exercise interference) |
Well-Known: GI upset, B12 deficiency risk, lactic acidosis (rare). |
| Senolytics (D+Q) | Physical Function: Improved 6-min walk distance and chair-stand speed in Idiopathic Pulmonary Fibrosis (Justice et al.) [10]. Tissue Clearance: Reduced senescent cell burden in adipose/skin of DKD patients [11]. |
Low (Small Pilot RCTs, n<20) | Unknown: "Hit-and-run" dosing tolerated in pilots; long-term risks defined. |
| NAD+ Boosters (NMN/NR) | Insulin Sensitivity: NMN improved muscle insulin sensitivity in prediabetic women (Yoshino et al.) [12]. Metabolism: NR raised NAD+ but showed no change in insulin sensitivity or body composition in healthy/obese men [13]. |
Moderate (Small RCTs) | High: Generally well-tolerated in trials to date. |
¶ Benefits
-
Caloric Restriction: The CALERIE-2 trial demonstrated that moderate caloric restriction is achievable in humans and significantly reduces cardiometabolic risk factors and the pace of biological aging [1:1][2:1].
-
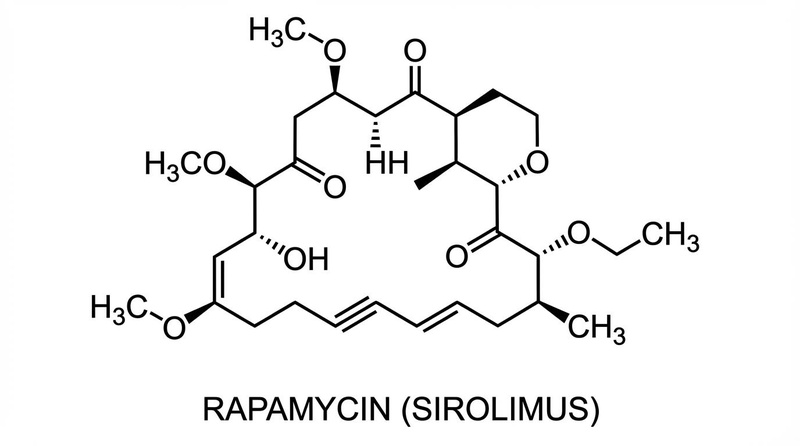
Rapamycin: While definitive life-extension data in humans is absent, mTOR inhibition has been shown to rejuvenate immune function in older adults, potentially reducing the burden of age-related infections [6:1].
-
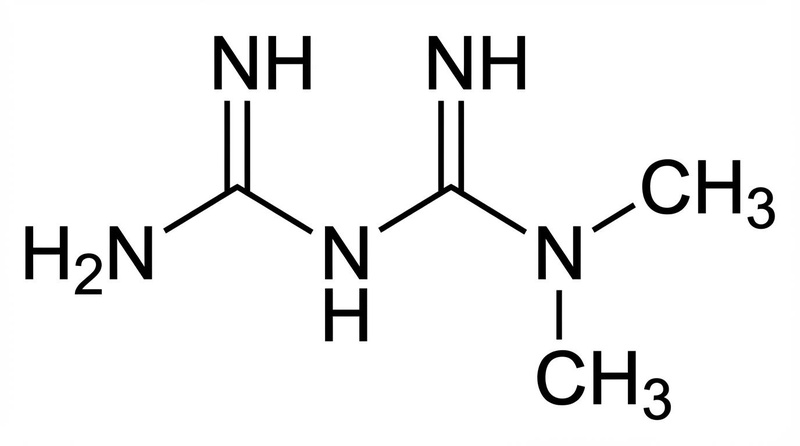
Metformin: Commonly used for diabetes, metformin has been linked to increased lifespan in observational studies of diabetics. However, caution is advised for healthy individuals engaging in exercise, as it may blunt the adaptive benefits of training [9:1].
-
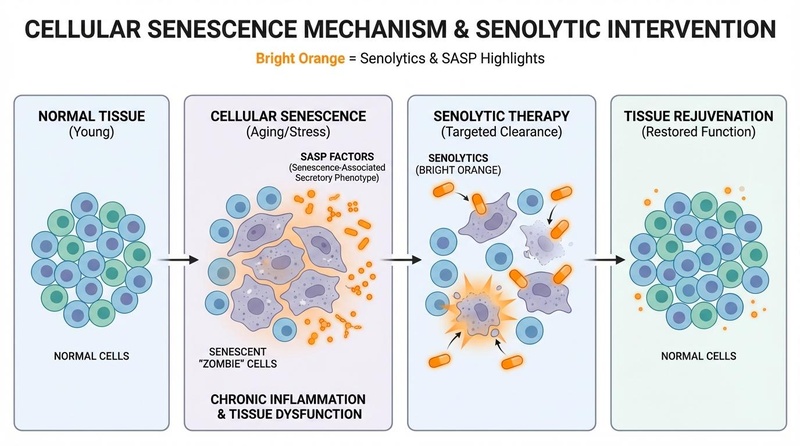
Senolytics: These agents target senescent cells ("zombie cells"). First-in-human pilot trials have demonstrated their ability to reduce senescent cell burden and improve physical function in specific disease states like pulmonary fibrosis [10:1].
-
NAD+ Boosters: NMN and NR reliably increase circulating NAD+ levels in humans. While metabolic benefits like improved insulin sensitivity have been seen with NMN in specific populations [12:1], broader anti-aging claims require more robust confirmation.
¶ Mechanism
These interventions target various biological pathways associated with aging (The Hallmarks of Aging).
- Caloric Restriction: Reduces metabolic stress, lowers core body temperature, and inhibits mTOR while activating AMPK.
- Rapamycin: Specific inhibitor of mTORC1, mimicking the signaling state of nutrient scarcity to enhance autophagy.
- Metformin: Activates AMPK and inhibits mitochondrial Complex I, improving insulin sensitivity.
- Senolytics: Temporarily disable pro-survival pathways (SCAPs) in senescent cells to induce apoptosis.
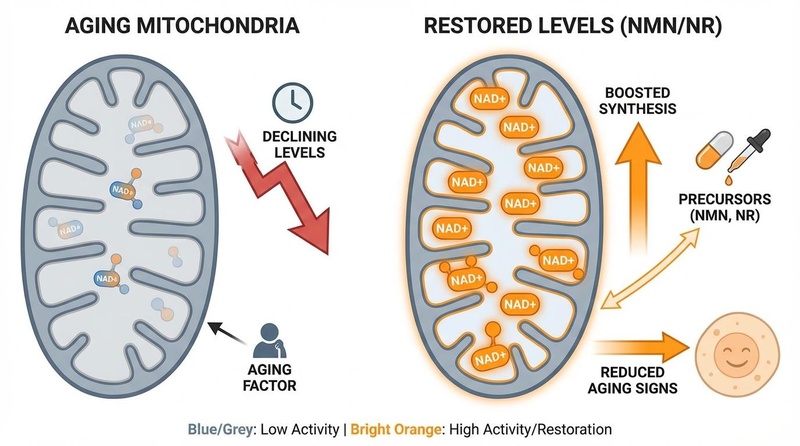
- NAD+ Boosters: Replenish declining NAD+ pools to fuel sirtuin activity (DNA repair) and mitochondrial function.
¶ Evidence
Recent systematic reviews and meta-analyses have highlighted the potential of these interventions:
- Caloric Restriction: Remains the gold standard intervention, with CALERIE-2 providing the most robust human evidence for healthspan extension [1:2].
- Rapamycin: Robust in mice (ITP); human evidence is currently limited to immune biomarkers and safety [6:2].
- Metformin: Strong evidence for diabetics; TAME trial is needed to determine efficacy in non-diabetics.
- Senolytics: Proof-of-concept achieved in humans; phase 2 trials are ongoing for various age-related indications.
- NAD+ Boosters: Bioavailability is solved; clinical efficacy for "rejuvenation" is still mixed and likely context-dependent [13:1].
¶ Safety & Side Effects
- Caloric Restriction: Potential risks include loss of libido, cold intolerance, and reduction in bone mineral density.
- Rapamycin: Side effects can include aphthous stomatitis (mouth ulcers), hyperlipidemia, and potential delayed wound healing.
- Metformin: GI discomfort (diarrhea/nausea) is common; may interfere with B12 absorption and exercise adaptations.
- Senolytics: Short-term "hit-and-run" protocols appear safe in pilots; long-term consequences of clearing senescent cells are unknown.
- NAD+ Boosters: Generally safe; some theoretical concerns about promoting growth of existing neoplasms exist but have not been observed in trials.
¶ FAQ
Q: Are these interventions safe?
A: Safety varies. Exercise and dietary interventions have high safety profiles. Pharmacological interventions like Rapamycin and Senolytics carry specific medical risks and should only be considered under clinical supervision.
Q: How long until we see definitive results?
A: Trials like TAME (Metformin) and PEARL (Rapamycin) are designed to provide more definitive answers on human aging in the coming years.
¶ References
Kraus WE, et al. 2 years of calorie restriction and cardiometabolic risk (CALERIE): exploratory outcomes of a multicentre, phase 2, randomised controlled trial. Lancet Diabetes Endocrinol. 2019;7(9):673-683. ↩︎ ↩︎ ↩︎
Belsky DW, et al. calorie restriction slows pace of aging in healthy adults (CALERIE). Nat Aging. 2022;2:105-116. ↩︎ ↩︎
Mandsager K, et al. Association of Cardiorespiratory Fitness With Long-term Mortality Among Adults Undergoing Exercise Treadmill Testing. JAMA Netw Open. 2018;1(6):e183605. ↩︎
Momma H, et al. Muscle-strengthening activities are associated with lower risk and mortality in major non-communicable diseases: a systematic review and meta-analysis of cohort studies. Br J Sports Med. 2022;56(13):755-763. ↩︎
Estruch R, et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet. N Engl J Med. 2018;378:e34. ↩︎
Mannick JB, et al. mTOR inhibition improves immune function in the elderly. Sci Transl Med. 2014;6(268):268ra179. ↩︎ ↩︎ ↩︎
Mannick JB, et al. TORC1 inhibition enhances immune function and reduces infections in the elderly. Sci Transl Med. 2018;10(449):eaaq1564. ↩︎
Campbell JM, et al. Metformin reduces all-cause mortality and diseases of ageing independent of its effect on diabetes control: A systematic review and meta-analysis. Ageing Res Rev. 2017;40:31-44. ↩︎
Konopka AR, et al. Metformin inhibits mitochondrial adaptations to aerobic exercise training in older adults. Aging Cell. 2019;18(1):e12880. ↩︎ ↩︎
Justice JN, et al. Senolytics in idiopathic pulmonary fibrosis: Results from a first-in-human, open-label, pilot study. EBioMedicine. 2019;40:229-234. ↩︎ ↩︎
Hickson LJ, et al. Senolytics decrease senescent cells in humans: Preliminary report from a clinical trial of Dasatinib plus Quercetin in individuals with diabetic kidney disease. EBioMedicine. 2019;47:446-456. ↩︎
Yoshino M, et al. Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women. Science. 2021;372(6547):1224-1229. ↩︎ ↩︎
Dollerup OL, et al. A randomized placebo-controlled clinical trial of nicotinamide riboside in obese men: safety, insulin-sensitivity, and lipid-mobilizing effects. Am J Clin Nutr. 2018;108(2):343-353. ↩︎ ↩︎