¶ Hyperbaric Oxygen Therapy
Hyperbaric oxygen therapy (HBOT) is a medical treatment that involves breathing 100% oxygen at elevated atmospheric pressure (typically 1.5-3.0 atmospheres absolute). While FDA-approved for specific conditions such as decompression sickness, carbon monoxide poisoning, and non-healing wounds[1], its application for longevity and age-related decline is investigational. Research is currently exploring HBOT's potential effects on cellular aging markers and physiological resilience.
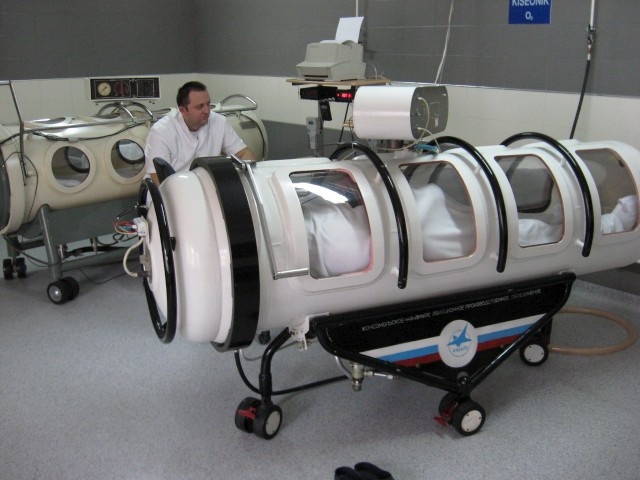
¶ Overview
By delivering high oxygen concentrations to tissues under increased pressure, HBOT creates a hyperoxic environment that may influence various biological responses. Emerging studies are investigating its effects on cellular aging markers, such as telomere dynamics, senescent cell burden, and mitochondrial function. The therapy is being evaluated as a potential approach to modulating age-related cellular processes, though its clinical efficacy and long-term safety for these specific applications remain under active research.
¶ Mechanisms
¶ Cellular and Molecular Pathways
HBOT is hypothesized to operate through several interconnected mechanisms relevant to aging biology:
Hypoxia-Inducible Factor (HIF) Activation: The intermittent hyperoxic-hypoxic cycling (created by air breaks or the end of sessions) activates HIF pathways, which may promote vascularization through vascular endothelial growth factor (VEGF) expression[2]. This angiogenic response could improve tissue oxygenation and support metabolic function.
Mitochondrial Enhancement: Enhanced oxygen availability may improve mitochondrial efficiency and cellular energy production[3]. This mechanism is proposed to address age-related mitochondrial dysfunction, a key hallmark of aging involved in cellular energy deficits.
Stem Cell Mobilization: HBOT has been shown to promote the release and activation of endogenous stem cells from bone marrow niches in some contexts[4]. These cells are involved in tissue repair and regeneration processes.
Modulation of Inflammation: Treatment appears to modulate inflammatory cytokines and may reduce chronic inflammation in certain pathological states[5]. Reducing chronic low-grade inflammation ("inflammaging") is a target for longevity interventions.
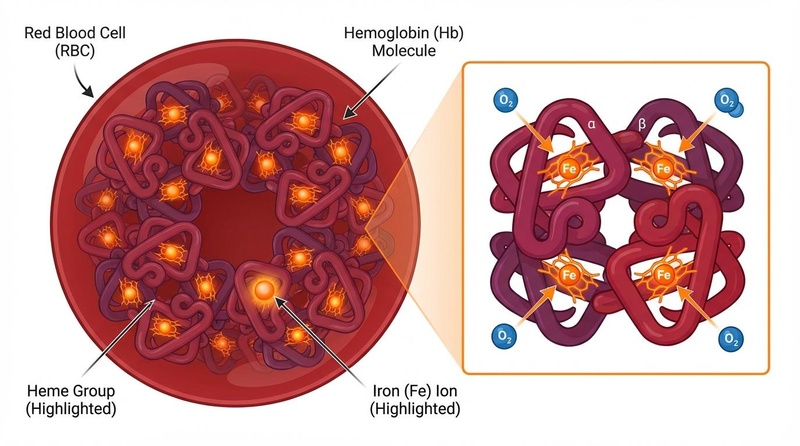
¶ Telomere and Senescence Biology
HBOT is being investigated for its potential to modify cellular aging markers, with preliminary findings suggesting:
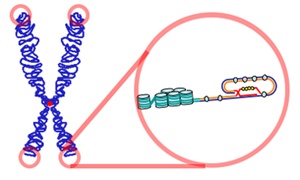
Telomere Elongation: A clinical trial reported 20-38% increases in telomere length in specific immune cell populations[6]. Telomere attrition is a primary hallmark of aging, and maintaining length is associated with cellular replicative capacity.
Senescent Cell Reduction: The same trial observed a reduction in senescent cell markers by 11-37% in immune cell subsets[6:1]. Senescent cells accumulate with age and secrete pro-inflammatory factors (SASP) that can damage surrounding tissues.
DNA Repair: Research suggests HBOT may activate cellular repair mechanisms through a hormetic stress response, potentially reducing DNA damage accumulation[7].
¶ Evidence
¶ Key Clinical Trial on Biomarkers (2020)
Hachmo et al. conducted a controlled study investigating HBOT's effects on aging biomarkers in humans[6:2]:
Study Design: 35 healthy adults aged 64+ underwent 60 HBOT sessions over 90 days (100% oxygen at 2 ATA with air breaks).
Reported Outcomes:
- Telomere length increased by 20-38% in B cells, T cells, and natural killer cells.
- Senescent cell populations decreased by 11-37% in specific immune cell subsets.
- These effects were observed on cellular markers; correlation with clinical longevity outcomes requires further study.
¶ Subsequent Research (2022-2024)
Recent studies have expanded the scope of HBOT longevity research:
Systematic Review (2024): A review of 15 studies on HBOT in aesthetic and aging-related applications reported improvements in skin aging biomarkers, such as collagen density and elastic fiber length, in several included studies[8].
Physical Performance Study (2024): A randomized controlled trial of 63 older adults reported significant improvements in VO2Max/kg (1.91 ± 3.29 ml/kg/min increase, p=0.0034) following 60 HBOT sessions, suggesting potential benefits for physical capacity[9].
Cognitive Function (2020): A separate RCT reported cognitive improvements in specific domains for healthy older adults using similar protocols, though clinical significance continues to be evaluated[10].
¶ Evidence Quality Assessment
| Outcome | Effect Size | Evidence Grade | Notes |
|---|---|---|---|
| Telomere length | +20-38% | C | Single RCT, n=35; biomarker outcome only[6:3] |
| Senescent cells | -11-37% | C | Flow cytometry markers; single RCT[6:4] |
| Physical performance | +1.9 ml/kg/min VO2Max | C | Single RCT, n=63[9:1] |
| Cognitive function | Small improvements | C | Limited to specific domains; single RCT[10:1] |
| Skin aging biomarkers | Increased collagen/elastic fibers | B | Systematic Review (15 studies)[8:1] |
Evidence grading: A (High - Multiple high-quality RCTs), B (Moderate - Several consistent RCTs), C (Low - Small/heterogeneous RCTs), D (Very Low - Observational/mechanistic).
¶ Safety and Contraindications
¶ Safety Profile
HBOT generally demonstrates a favorable safety profile when administered in clinical settings under medical supervision. Adverse event rates are approximately 0.4% in established clinical use[11]. However, risks exist, particularly with improper pressurization or unsupervised use.
¶ Common Side Effects
Barotrauma (most frequent, ~50% of adverse events):
- Ear pain or discomfort during pressure changes
- Sinus pressure or congestion
- Temporary hearing changes
General Effects:
- Post-treatment fatigue (typically resolves quickly)
- Temporary lightheadedness
- Claustrophobia or confinement anxiety (~25% of adverse events)
Vision Changes:
- Transient myopia (nearsightedness) lasting 6-8 weeks post-treatment
- Generally reversible, though cataract maturation can theoretically be accelerated in some cases.
¶ Contraindications
Established guidelines list specific absolute and relative contraindications for HBOT[12]:
Absolute Contraindications:
- Untreated pneumothorax (risk of tension pneumothorax)
- Current use of doxorubicin or cisplatin (chemotherapy agents)
- Intraocular gas bubbles
Relative Contraindications (require careful medical evaluation):
- Chronic obstructive pulmonary disease (COPD) with bullae
- Active asthma symptoms
- Severe claustrophobia
- Recent ear or sinus surgery
- Pregnancy (precautionary)
- Seizure history (oxygen toxicity risk)
¶ Monitoring Requirements
Pre-treatment Assessment:
- Comprehensive medical history
- Ear, nose, and throat examination
- Glucose monitoring for diabetic patients
- Review of medications for potential interactions
During Treatment:
- Blood glucose monitoring (can drop during HBOT)
- Ear equalization assessment
- Vital signs monitoring
¶ Practical Considerations
¶ Treatment Protocols
Standard Research Protocol (based on longevity trials):
- Pressure: 2.0 ATA (atmospheres absolute)
- Duration: 90 minutes per session
- Frequency: 5 sessions per week
- Total: 60 sessions over 12 weeks
- Oxygen: 100% via mask with periodic air breaks (to trigger HIF response)
Note: "Soft" hyperbaric chambers (typically 1.3 ATA, no 100% oxygen concentration) operate differently and evidence from 2.0 ATA trials may not apply to these devices.
¶ Cost and Accessibility
Financial Considerations:
- Individual sessions: $200-500 per treatment in clinical centers
- Complete protocol (60 sessions): $12,000-30,000
- Insurance typically excludes longevity, wellness, or "anti-aging" applications
- "Soft" chambers are cheaper but may not provide the same physiological stimulus
Facility Requirements:
- Trained medical supervision is mandatory for high-pressure (medical) HBOT
- Proper chamber certification (PVHO) and fire suppression systems
- Emergency protocols
¶ Clinical Applications
¶ Approved Medical Uses
HBOT has FDA approval for specific medical conditions, including but not limited to:
- Decompression sickness
- Carbon monoxide poisoning
- Non-healing diabetic wounds
- Delayed radiation injury
- Gas gangrene
¶ Investigational Longevity Applications
Use of HBOT for longevity is considered investigational and off-label. Areas of active research include:
- Modification of cellular aging biomarkers (telomeres, senescence)
- Enhancement of physical and cognitive performance in healthy aging
- Reduction of systemic inflammation
¶ Regulatory Status
FDA Classification: Approved for specific medical indications only. The FDA has issued warnings against misleading claims regarding HBOT for anti-aging, autism, or cancer cures where evidence is lacking[1:1].
Clinical Guidelines: No standardized clinical guidelines exist for longevity applications.
Research Status: Early-phase trials have shown biomarker changes; larger, long-term Phase III studies are needed to establish clinical benefits for aging.
¶ Future Prospects
¶ Technological Developments
Home-based Systems: Research is ongoing into whether lower-pressure protocols (amenable to home use) can elicit meaningful physiological responses, though current evidence favors higher pressures (2.0 ATA) for stem cell and telomere effects.
Protocol Optimization: Efforts to define the "minimum effective dose" for longevity benefits to reduce the time and cost burden.
¶ Research Priorities
Large-scale Trials: Multi-center randomized controlled trials are needed to validate preliminary biomarker findings.
Clinical Correlation: Establishing whether observed changes in telomeres and senescent cells translate to actual extended healthspan or lifespan in humans.
Safety Optimization: Long-term monitoring of cumulative oxygen exposure in healthy individuals.
¶ Conclusion
Hyperbaric oxygen therapy is an established medical treatment for specific injuries and conditions. Its application for longevity is an area of active investigation, with some clinical trials reporting positive effects on cellular aging markers like telomere length and senescent cell burden. However, these applications are not FDA-approved, and the long-term clinical significance of these biomarker changes remains to be fully established. Patients considering HBOT for longevity should distinguish between proven medical uses and experimental wellness protocols.
¶ References
Food and Drug Administration. Hyperbaric oxygen therapy: don't be misled. 2024. https://www.fda.gov/consumers/consumer-updates/hyperbaric-oxygen-therapy-dont-be-misled ↩︎ ↩︎
Hadanny A, Efrati S. The hyperoxic-hypoxic paradox. Biomolecules. 2020;10(6):958. https://pubmed.ncbi.nlm.nih.gov/32575704/ ↩︎
Thom SR. Hyperbaric oxygen: its mechanisms and efficacy. Plast Reconstr Surg. 2011;127 Suppl 1:131S-141S. https://pubmed.ncbi.nlm.nih.gov/21200281/ ↩︎
Thom SR, Bhopale VM, Velazquez OC, et al. Stem cell mobilization by hyperbaric oxygen. Am J Physiol Heart Circ Physiol. 2006;290(4):H1378-1386. https://pubmed.ncbi.nlm.nih.gov/16293654/ ↩︎
Efrati S, Hadanny A. Hyperbaric oxygen therapy for healthy aging: From mechanisms to therapeutics. Mech Ageing Dev. 2022;206:111695. https://pubmed.ncbi.nlm.nih.gov/35649339/ ↩︎
Hachmo Y, Hadanny A, Mendelovic S, et al. Hyperbaric oxygen therapy increases telomere length and decreases immunosenescence in isolated blood cells: a prospective trial. Aging (Albany NY). 2020;12(22):22445-22456. https://pubmed.ncbi.nlm.nih.gov/33206062/ ↩︎ ↩︎ ↩︎ ↩︎ ↩︎
Godman CA, Chheda KP, Hightower LE, et al. Hyperbaric oxygen induces a cytoprotective and angiogenic response in human microvascular endothelial cells. Cell Stress Chaperones. 2010;15(4):431-442. https://pubmed.ncbi.nlm.nih.gov/20623200/ ↩︎
Fisher SM, Sherif RD, Borab ZM, Kumar NG, Rohrich RJ. Hyperbaric oxygen therapy in aesthetic medicine and anti-aging: A systematic review. Aesthet Plast Surg. 2024;49:2534–2544. https://pubmed.ncbi.nlm.nih.gov/39733047/ ↩︎ ↩︎
Hadanny A, Sasson E, Copel L, et al. Physical enhancement of older adults using hyperbaric oxygen: a randomized controlled trial. BMC Geriatr. 2024;24(1):572. https://pubmed.ncbi.nlm.nih.gov/38942582/ ↩︎ ↩︎
Hadanny A, Daniel-Kotovsky M, Suzin G, et al. Cognitive enhancement of healthy older adults using hyperbaric oxygen: a randomized controlled trial. Aging (Albany NY). 2020;12(13):13740-13761. https://pubmed.ncbi.nlm.nih.gov/32663205/ ↩︎ ↩︎
Heyboer M 3rd, Sharma D, Santiago W, et al. Hyperbaric oxygen therapy: side effects defined and quantified. Adv Wound Care (New Rochelle). 2017;6(6):210-224. https://pubmed.ncbi.nlm.nih.gov/28509569/ ↩︎
Weaver LK. Hyperbaric oxygen therapy indications. 13th ed. North Palm Beach, FL: Best Publishing Company; 2014. https://www.bestpub.com/books/hyperbaric-and-undersea-medicine/product/hyperbaric-oxygen-therapy-indications-13th-edition.html ↩︎