¶ Nicotinamide Mononucleotide (NMN)
Human trials up to 1250 mg/day have shown no serious adverse events.
Caution: Theoretical risk of methyl-group depletion; often paired with TMG.
¶ Standard Protocol
- Dosage (General): 300–600 mg daily.
- Dosage (Athletic/Therapeutic): Up to 900–1000 mg daily has been used in trials for aerobic capacity.
- Formulation: Liposomal delivery is preferred to protect NMN from stomach acid and gut bacteria. Sublingual powder is the second-best option to bypass first-pass metabolism.
- Timing: Take in the morning (NAD+ levels follow a circadian rhythm).
- Synergy: Often stacked with Trimethylglycine (TMG) (500–1000 mg) to support methylation, and Resveratrol or Pterostilbene to activate Sirtuins.
- Storage: Must be kept cold (refrigerated) if not stabilized or liposomal. NMN degrades into Nicotinamide (NAM) in heat/humidity.
¶ Bottom Line
NMN is currently the most popular intervention for restoring declining NAD+ levels. Unlike its precursor Nicotinamide Riboside (NR), NMN has demonstrated specific benefits for muscle insulin sensitivity and aerobic capacity in humans. Following a multi-year regulatory battle, the FDA confirmed its lawful status as a dietary supplement in late 2025.
¶ Benefits & Applications
¶ Metabolic Health & Insulin Sensitivity
NMN has shown a unique ability to improve how skeletal muscle processes glucose. In a landmark 2021 study, postmenopausal women with prediabetes who took 250 mg of NMN daily for 10 weeks saw a 25% increase in muscle insulin sensitivity[1]. This effect is comparable to some prescription therapeutics and significant weight loss.
¶ Physical Performance & Aerobic Capacity

Recent trials indicate NMN can enhance endurance. Amateur runners taking 600–1200 mg/day showed improved oxygen uptake (VO2) and power output at ventilatory thresholds[2]. In older adults, daily supplementation has been shown to improve six-minute walking distance, a key metric of functional mobility[3].
¶ Cellular Energy & Repair
By boosting NAD+, NMN fuels:
- Mitochondrial Function: Improving ATP production for physical and mental energy.
- DNA Repair: Supporting PARP enzymes that fix genomic damage.
- Sirtuin Activation: Fueling the "longevity genes" (SIRT1–7) that regulate inflammation and stress resistance.
¶ The Reality Check
¶ 1. The "FDA Saga" (2022–2025)
NMN's availability has been volatile due to regulatory actions.
- The Ban (2022/2023): In late 2022, the FDA declared NMN a "drug" (due to an Investigational New Drug application by Metro International Biotech) and excluded it from being a supplement. Amazon subsequently banned sales in March 2023.
- The Reversal (2025): Following a lawsuit by the Natural Products Association (NPA), the FDA reversed its position on September 29, 2025, confirming that NMN is lawful for use in dietary supplements, citing evidence that it was marketed as a supplement prior to the drug authorization[4][5].
- Current Status: NMN is legal and widely available in the US market again.
¶ 2. Bioavailability: The "Gut Barrier"
Standard oral NMN is largely unstable in the gut. Stable isotope studies suggest that a significant portion of oral NMN is deamidated by gut bacteria into Nicotinamide (NAM) before absorption, then resynthesized into NAD+ in the liver[6].
- Liposomal Advantage: A 2025 head-to-head trial found that Liposomal NMN increased blood NAD+ levels by 84% compared to standard powder, likely by protecting the molecule from gastric and bacterial degradation[7].
¶ 3. Storage Matters
NMN is sensitive to heat and moisture. In hot/humid conditions, it degrades into Nicotinamide, which can actually inhibit Sirtuins at high concentrations. Unless you are buying a specifically stabilized form (often crystalline) or liposomal product, store your NMN in the fridge.
¶ Evidence Room
¶ Mechanism of Action
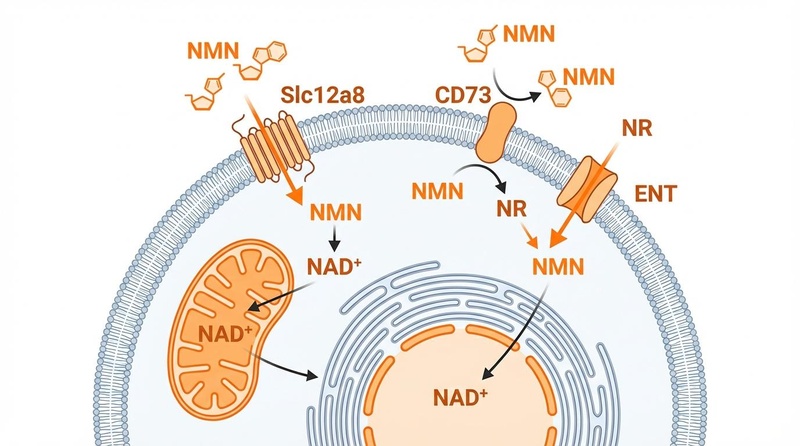
NMN enters cells to replenish the NAD+ pool, which declines steadily with age (dropping by ~50% between age 20 and 60).
- Entry Routes: NMN can enter cells directly via the Slc12a8 transporter (highly expressed in the gut) or be converted extracellularly to NR by CD73, enter via ENT transporters, and be re-phosphorylated to NMN by NRK enzymes[8].
- Downstream Effects: Once inside, NMN is converted to NAD+ by NMNAT enzymes. This NAD+ is then consumed by Sirtuins (deacetylases) and PARPs (DNA repair), producing Nicotinamide (NAM) as a byproduct, which is recycled back into NMN (Salvage Pathway).
¶ Evidence Summary Table (Human Outcomes)
| Study | Participants | Dose/Duration | Primary Outcome | Certainty (GRADE) |
|---|---|---|---|---|
| Yoshino et al. (2021)[1:1] | 25 Prediabetic Women | 250 mg/day (10 wks) | ↑ 25% Muscle Insulin Sensitivity (p<0.01) | High |
| Yi et al. (2023)[3:1] | 80 Healthy Adults (40-65y) | 300–900 mg/day (60 days) | ↑ Blood NAD+ (p<0.001) ↑ 6-min Walking Distance (p<0.01) |
Moderate |
| Liao et al. (2021)[2:1] | 48 Runners | 300–1200 mg/day (6 wks) | ↑ Aerobic Capacity (VO2) (Dose-dependent, p<0.05) | Moderate |
| Kawakami et al. (2025)[7:1] | 15 Healthy Men | 350 mg Liposomal vs Std | Liposomal ↑ NAD+ by 84% vs Standard (p=0.001) | Low (Small sample) |
| Igarashi et al. (2022)[9] | 42 Older Men | 250 mg/day (12 wks) | ↑ NAD+; No change in grip strength or visceral fat | Moderate |
¶ Safety & Toxicology
- Methylation: High-dose NMN (like NR) requires methyl groups for the excretion of its byproduct, Nicotinamide. While trials up to 1000 mg haven't shown dangerously elevated homocysteine, long-term depletion of the methyl pool is a theoretical risk. Recommendation: Co-supplement with TMG (Trimethylglycine).
- Tumor Promotion: Because tumors consume NAD+, there is a theoretical fear that NMN could fuel cancer growth. However, animal studies (e.g., Pan et al., 2021) in lung cancer models showed no promotion of tumor growth with NMN[10].
- Maximum Tolerated Dose: Human safety has been established up to 1250 mg/day.
¶ Comparison: NMN vs. Nicotinamide Riboside (NR)
- Efficacy: Both effectively raise NAD+. A 2026 head-to-head comparison suggested NR might raise blood NAD+ slightly higher (2.3x vs 1.7x) in the short term[11], but NMN has stronger data for insulin sensitivity (which NR trials have failed to show).
- Stability: NR (as Chloride) is shelf-stable; NMN requires cold storage unless liposomal/stabilized.
- Transport: NMN uses Slc12a8; NR uses ENT transporters.
¶ References
Yoshino, M., et al. (2021). Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women. Science, 372(6547), 1224–1229. https://doi.org/10.1126/science.abe9985 ↩︎ ↩︎
Liao, B., et al. (2021). Nicotinamide mononucleotide supplementation enhances aerobic capacity in amateur runners: a randomized, double-blind study. Journal of the International Society of Sports Nutrition, 18, 54. https://doi.org/10.1186/s12970-021-00442-4 ↩︎ ↩︎
Yi, L., et al. (2023). The efficacy and safety of β-nicotinamide mononucleotide (NMN) supplementation in healthy middle-aged adults: a randomized, multicenter, double-blind, placebo-controlled, parallel-group, dose-dependent clinical trial. GeroScience, 45, 29–43. https://doi.org/10.1007/s11357-022-00705-1 ↩︎ ↩︎
U.S. Food and Drug Administration. (2025, September 29). Response to Citizen Petition from Natural Products Association regarding NMN. https://www.npanational.org/news/fda-reinstates-nmn-as-dietary-supplement-after-npa-lawsuit/ ↩︎
CIRS Group. (2025). US FDA Confirms NMN Lawful in Dietary Supplements. https://www.cirs-group.com/en/food/us-fda-confirms-nmn-lawful-in-dietary-supplements ↩︎
Christen, S., et al. (2025). A randomized, open-label, placebo-controlled study to evaluate the effects of three NAD+ precursors on NAD+ levels and gut health in healthy adults. Nature Metabolism. https://www.nature.com/natmetab/ ↩︎
Kawakami, S., et al. (2025). Intervention Study Comparing Blood NAD+ Concentrations with Liposomal and Non-Liposomal Nicotinamide Mononucleotide. Annals of Clinical and Medical Case Reports, 14(11). https://acmcasereport.org/ ↩︎ ↩︎
Grozio, A., et al. (2019). Slc12a8 is a nicotinamide mononucleotide transporter. Nature Metabolism, 1, 47–57. https://doi.org/10.1038/s42255-018-0009-4 ↩︎
Igarashi, M., et al. (2022). Chronic nicotinamide mononucleotide supplementation elevates blood nicotinamide adenine dinucleotide levels and alters muscle motility in healthy old men. NPJ Aging, 8, 5. https://doi.org/10.1038/s41514-022-00084-z ↩︎
Pan, F., et al. (2021). Effect of β-nicotinamide mononucleotide on tumor formation and growth in a lung cancer mouse model. Materials Chemistry Frontiers, 5, 995-1002. https://doi.org/10.1039/D0QM00897D ↩︎
Berven, H., et al. (2026). NR-SAFE: a randomized, double-blind safety trial of high dose nicotinamide riboside in Parkinson's disease. Nature Communications, 14, 7793. https://doi.org/10.1038/s41467-023-43514-6 ↩︎