¶ Rapamycin for Longevity and Anti-Aging
The "Gold Standard" of pharmacological lifespan extension.
¶ Overview

Rapamycin (sirolimus) is a macrolide compound originally discovered in the soil of Easter Island (Rapa Nui) in 1972. Initially developed as an antifungal agent, it was later FDA-approved as an immunosuppressant for organ transplant recipients to prevent rejection. Over the last two decades, it has emerged as the premier candidate for pharmacological lifespan extension, demonstrating robust and reproducible effects across diverse species.
It is currently the only drug consistently proven to extend lifespan in both male and female mice across multiple independent sites in the National Institute on Aging's Interventions Testing Program (ITP). Its mechanism centers on the inhibition of the mechanistic Target of Rapamycin (mTOR), a conserved serine/threonine kinase that functions as a master regulator of cell growth, metabolism, and aging.
¶ Mechanism of Action
The anti-aging effects of rapamycin are mediated through its precise modulation of the mTOR signaling network. This pathway integrates nutrient availability (amino acids, glucose), growth factors, and cellular stress to dictate the cellular switch between anabolism (growth) and catabolism (repair/autophagy).
¶ The mTOR Complexes: mTORC1 vs. mTORC2
mTOR operates in two distinct protein complexes, which differ in function and sensitivity to rapamycin:
- mTORC1 (Rapamycin-Sensitive): This complex acts as the "growth switch." It is the primary regulator of protein synthesis, ribosome biogenesis, and cell proliferation. It is acutely sensitive to rapamycin inhibition.
- mTORC2 (Rapamycin-Insensitive): This complex regulates cytoskeletal organization and insulin signaling (via AKT phosphorylation). It is generally insensitive to acute rapamycin treatment, though chronic exposure can inhibit it indirectly.
¶ Molecular Specificity
Rapamycin acts as an allosteric inhibitor. It does not bind directly to the kinase active site; instead, it binds to the intracellular protein FKBP12. This Rapamycin-FKBP12 complex then binds to the FRB domain of mTORC1, destabilizing it.
- Effect: Inhibition of mTORC1 suppresses the phosphorylation of downstream effectors S6K1 (which drives protein synthesis) and 4E-BP1 (which regulates translation initiation)[1].
- Result: The cell shifts from a "growth" state to a "maintenance" state.
¶ Autophagy Induction
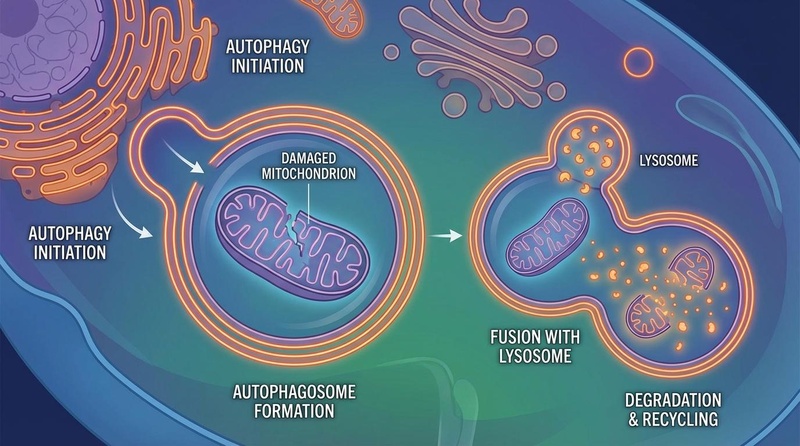
A critical downstream effect of mTORC1 inhibition is the induction of autophagy (cellular recycling). Under nutrient-rich conditions, mTORC1 inhibits the ULK1 complex, blocking autophagy. Rapamycin releases this brake, allowing cells to clear damaged organelles (mitophagy) and misfolded proteins, thereby preserving proteostasis and preventing the accumulation of cellular debris associated with aging[2].