¶ Nicotinamide Riboside

| Type | Vitamin B3 / Nucleoside |
| Active Cmpd | Nicotinamide Riboside |
| Source | Milk (trace), Yeast, Synthetic |
| Dose Range | 300 – 1,000 mg/day |
| Half-life | ~2.7 hours (plasma) |
| Main Benefit | Restores NAD+ Levels |
| Absorption | High (Oral) |
A potent NAD+ precursor and form of Vitamin B3 that safely elevates cellular NAD+ levels in humans. While it shows promise for neuroprotection and rare age-related diseases, evidence for general metabolic or physical performance benefits in healthy adults is currently mixed.
¶ At a glance
Aliases
- NR
- Niagen (Patented Chloride form)
- 1-(β-D-Ribofuranosyl)nicotinamide
Key points
- Strongest Effect: Consistently doubles blood NAD+ levels in humans with oral supplementation (1,000 mg/day)[1][2].
- Clinical Promise: Shows potential in neurodegenerative conditions (Parkinson’s, ALS) and rare genetic aging disorders (Werner Syndrome)[3][4].
- Key Limitation: Unlike in mice, human trials have largely failed to show improvements in insulin sensitivity, body composition, or exercise performance in healthy or obese adults[5][6].
- Safety: Excellent safety profile in trials up to 2 years; does not cause the "flushing" associated with Niacin[7].
What people use it for
- Restoring NAD+ levels (anti-aging)
- Neuroprotection (Parkinson’s, cognitive resilience)
- Mitochondrial support
- Evidence quality (NAD+ elevation): High
- Evidence quality (Clinical outcomes): Low to Moderate
¶ What is Nicotinamide Riboside?
Nicotinamide Riboside (NR) is a form of Vitamin B3 that acts as a precursor to Nicotinamide Adenine Dinucleotide (NAD+), a critical coenzyme found in every living cell. NAD+ is essential for converting food into energy (ATP) and for fueling enzymes like sirtuins and PARPs that repair DNA and regulate cellular aging.
Unlike other forms of Vitamin B3 (like niacin or nicotinamide), NR uses a unique pathway to enter cells and convert to NAD+, bypassing the rate-limiting enzyme NAMPT. This allows it to efficiently boost NAD+ levels even when normal synthesis pathways are compromised by age or stress.
Commercial Forms:
- NR Chloride (Niagen): The most stable and widely studied form, used in almost all human clinical trials.
- NR Trioleate: An oil-soluble form designed for liquid stability, currently less common.
¶ What are Nicotinamide Riboside's main benefits?
¶ 1. Consistent Restoration of NAD+ Levels
The most definitive benefit of NR is its ability to raise NAD+ levels. Multiple human trials have confirmed that daily supplementation (typically 1,000 mg) increases whole-blood and tissue NAD+ by approximately 100% (2-fold) within 2 weeks, maintaining these levels with continued use[1:1][2:1].
¶ 2. Neuroprotection and Brain Health
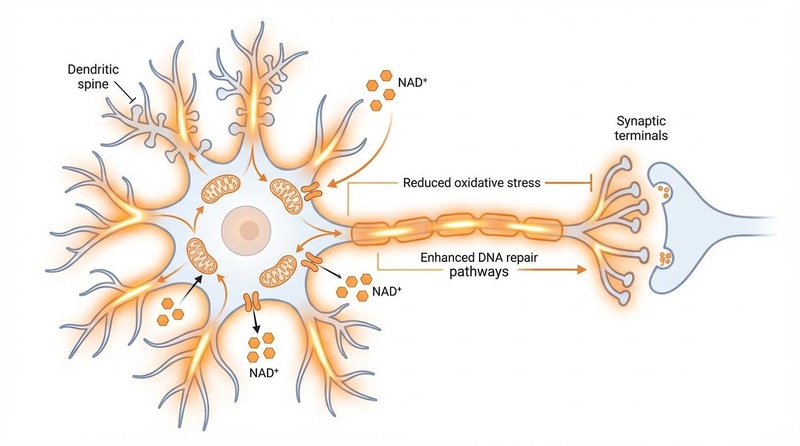
Research suggests NR may cross the blood-brain barrier and support neuronal health.
- Parkinson's Disease: The NADPARK trial (Phase I) showed that NR (1,000 mg) increased cerebral NAD+ levels and was associated with mild clinical improvements in Parkinson's patients[3:1].
- ALS: A pilot study indicated that NR combined with pterostilbene might slow progression in ALS patients, though larger confirmatory trials are ongoing[8].
¶ 3. Rare Aging Disorders (Werner Syndrome)
In a landmark 2025 study from Chiba University, NR supplementation was found to improve arterial stiffness and reduce skin ulcers in patients with Werner Syndrome, a genetic disorder characterized by rapid, premature aging. This provides "proof of concept" for NR's ability to mitigate specific age-related pathologies in humans[4:1].
¶ 4. Anti-Inflammatory Effects
Some studies indicate NR may reduce circulating inflammatory cytokines (like IL-6 and IL-5) in older adults, potentially modulating "inflammaging," although this has not yet translated into clear functional improvements in healthy people[9].
¶ Evidence summary table (human outcomes)
| Outcome / Goal | Effect | Consistency | Evidence quality | Trials | Notes |
|---|---|---|---|---|---|
| Blood NAD+ Levels | High | High | >10 RCTs | Consistently doubles levels at 1g/day[1:2][2:2] | |
| Cerebral NAD+ | High | Moderate | 2 RCTs | Increased in Parkinson's patients[3:2] | |
| Insulin Sensitivity | High | Moderate | 3 RCTs | No improvement in obese/insulin-resistant men[5:1][6:1] | |
| Body Composition | Low | Low | 1 RCT | Slight ↑ in lean mass in one study; others null[10] | |
| Exercise Performance | High | Moderate | 2 RCTs | No benefit in healthy young men[11] | |
| Blood Pressure | Moderate | Low | 1 RCT | Reduction seen only in those with elevated baseline BP[12] | |
| Werner Syndrome Sx | Moderate | Moderate | 1 RCT | Improved skin ulcers and arterial stiffness (2025)[4:2] |
¶ How does Nicotinamide Riboside work?
¶ The "Hidden" Pathway
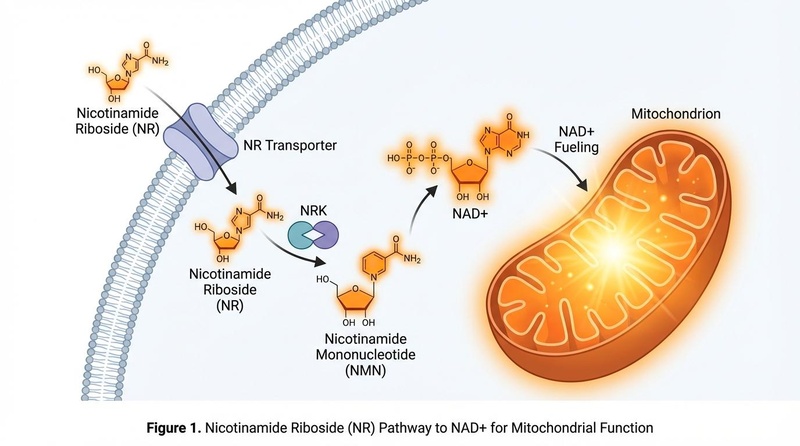
NR enters cells via Equilibrative Nucleoside Transporters (ENTs). Once inside, it is phosphorylated by specific enzymes called NR Kinases (NRK1/2) to become Nicotinamide Mononucleotide (NMN), which is then quickly converted to NAD+.
This "NRK pathway" is often upregulated when cells are under metabolic stress, making NR a particularly effective rescue molecule for failing cells.
¶ NR vs. NMN: The Transport Debate
- NR: Enters via ENTs, which are ubiquitous (found in almost all cells).
- NMN: Requires a specific transporter (Slc12a8) found primarily in the gut and certain tissues. Some research suggests NMN may need to be converted into NR outside the cell (by losing a phosphate group) to enter, before being turned back into NMN inside the cell[13].
- Practical Outcome: Both precursors ultimately feed the same pool of NAD+, and head-to-head human data suggests they are similarly effective at raising blood NAD+ levels.
¶ Dosage and how to take it
¶ Standard Dosing
- Clinical Trials: Most efficacy studies use 1,000 mg per day, often split into two doses (500 mg morning, 500 mg evening).
- Consumer Maintenance: Many users take 300 mg per day for general health, though this dose produces a smaller rise in NAD+ than the 1,000 mg protocol used in major trials.
¶ Forms and Bioavailability
- NR Chloride (Niagen): The gold standard for stability and absorption.
- Timing: Can be taken with or without food. Taking it with a meal (especially breakfast) may help mitigate mild nausea if it occurs.
¶ Safety and side effects
NR is considered one of the safest longevity supplements, with trials administering up to 2,000 mg/day for 12 weeks and 1,000 mg/day for up to 2 years without serious adverse events.
Common Side Effects
- Mild nausea or GI upset (rare, dose-dependent).
- Headaches (transient).
- No Flushing: unlike Niacin, NR does not cause skin flushing.
Who should be cautious
- Cancer: Theoretical concern that boosting NAD+ could fuel rapidly dividing cancer cells. While no human data supports this risk, clinical guidance typically advises caution in active cancer cases due to these theoretical mechanisms.
¶ Comparison: NR vs. Other Forms
| Feature | Nicotinamide Riboside (NR) | Nicotinamide Mononucleotide (NMN) | Niacin (NA) |
|---|---|---|---|
| NAD+ Elevation | High (~2x) | High (~2x) | High |
| Cell Entry | Direct (ENTs) | Slc12a8 or conversion to NR | GPR109A |
| Side Effects | None/Mild GI | None/Mild GI | Severe Flushing |
| Human Evidence | High (Parkinson's, ALS, Werner's) | Moderate (Insulin sensitivity) | High (Lipids/Cholesterol) |
| Regulatory | Legal Supplement (US/EU) | Complex (Drug exclusion in US) | Legal Supplement |
¶ References
Airhart SE, et al. An open-label, non-randomized study of the pharmacokinetics of the nutritional supplement nicotinamide riboside (NR) and its effects on blood NAD+ levels in healthy volunteers. PLoS One. 2017. https://doi.org/10.1371/journal.pone.0186459 ↩︎ ↩︎ ↩︎
Martens CR, et al. Chronic nicotinamide riboside supplementation is well-tolerated and elevates NAD+ in healthy middle-aged and older adults. Nat Commun. 2018. https://doi.org/10.1038/s41467-018-03421-7 ↩︎ ↩︎ ↩︎
Brakedal B, et al. The NADPARK study: A randomized phase I trial of nicotinamide riboside supplementation in Parkinson's disease. Cell Metab. 2022. https://doi.org/10.1016/j.cmet.2022.02.001 ↩︎ ↩︎ ↩︎
Shoji N, et al. Nicotinamide riboside supplementation benefits in patients with Werner syndrome: A double-blind randomized crossover placebo-controlled trial. Aging Cell. 2025. https://doi.org/10.1111/acel.70093 ↩︎ ↩︎ ↩︎
Dollerup OL, et al. A randomized placebo-controlled clinical trial of nicotinamide riboside in obese men: safety, insulin-sensitivity, and lipid-mobilizing effects. Am J Clin Nutr. 2018. https://doi.org/10.1093/ajcn/nqy132 ↩︎ ↩︎
Dollerup OL, et al. Effects of Nicotinamide Riboside on Endocrine Pancreatic Function and Incretin Hormones in Nondiabetic Men With Obesity. J Clin Endocrinol Metab. 2019. https://doi.org/10.1210/jc.2019-01081 ↩︎ ↩︎
Conze D, et al. Safety and Metabolism of Long-term Administration of NIAGEN (Nicotinamide Riboside Chloride) in a Randomized, Double-Blind, Placebo-controlled Clinical Trial of Healthy Overweight Adults. Sci Rep. 2019. https://doi.org/10.1038/s41598-019-46120-z ↩︎
De la Rubia JE, et al. Efficacy and tolerability of EH301 for amyotrophic lateral sclerosis: a randomized, double-blind, placebo-controlled human pilot study. Amyotroph Lateral Scler Frontotemporal Degener. 2019. https://doi.org/10.1080/21678421.2018.1536152 ↩︎
Elhassan YS, et al. Nicotinamide Riboside Augments the Aged Human Skeletal Muscle NAD+ Metabolome and Induces Transcriptomic and Anti-inflammatory Signatures. Cell Rep. 2019. https://doi.org/10.1016/j.celrep.2019.07.043 ↩︎
Remie CME, et al. Nicotinamide riboside supplementation alters body composition and skeletal muscle acetylcarnitine concentrations in healthy obese humans. Am J Clin Nutr. 2020. https://doi.org/10.1093/ajcn/nqaa134 ↩︎
Stocks B, et al. Nicotinamide riboside supplementation does not alter whole-body or skeletal muscle metabolic responses to a single bout of endurance exercise. J Physiol. 2021. https://doi.org/10.1113/JP280825 ↩︎
Martens CR, et al. Chronic nicotinamide riboside supplementation is well-tolerated and elevates NAD+ in healthy middle-aged and older adults. Nat Commun. 2018. https://doi.org/10.1038/s41467-018-03421-7 ↩︎
Grozio A, et al. Slc12a8 is a nicotinamide mononucleotide transporter. Nat Metab. 2019. https://doi.org/10.1038/s42255-018-0009-4 ↩︎