¶ NAD+ (Nicotinamide Adenine Dinucleotide)
¶ At a Glance
Definition: NAD+ is a universal electron carrier and enzymatic cofactor found in every living cell. It is the "currency of metabolism," essential for converting nutrients into energy (ATP) and acting as a consumed fuel for longevity proteins.
The Aging Crisis: NAD+ levels decline by up to 50% between young adulthood and old age. This collapse is a primary driver of mitochondrial dysfunction, genomic instability, and cellular senescence.[1]
Biological Role:
- Energy: Without NAD+, mitochondrial energy production stops instantly.
- Signaling: It is the "on switch" for Sirtuins (longevity genes) and DNA repair enzymes.
Intervention Strategy: NAD+ itself has poor oral bioavailability and cannot easily cross cell membranes. Clinical strategies focus on precursors (like NMN and NR) that the cell can convert into NAD+.
👉 For intervention protocols, dosage, and supplements, see NAD+ Precursors.
¶ The "Why": Fuel for Life
NAD+ exists in two distinct forms, and the ratio between them determines the metabolic state of the cell.
¶ 1. The Redox Engine (NADH)
In its primary role, NAD+ acts as an electron shuttle. It accepts high-energy electrons from the breakdown of glucose and fatty acids to become NADH (reduced form). It then donates these electrons to the Electron Transport Chain in the mitochondria to generate ATP.
- The Cycle: In young, healthy cells, NAD+ is constantly recycled back and forth to NADH. This functions as a catalyst—it is not consumed, just reused.
¶ 2. The Consumable Signaling Molecule
In its secondary role—critical for aging—NAD+ is permanently consumed by enzymes. It is broken down to power reactions that repair DNA and regulate gene expression.
- The Conflict: Because these enzymes destroy NAD+, they compete with the mitochondria for the limited pool. If consumption exceeds the cell's ability to make more (synthesis), the entire system crashes.
¶ Mechanisms of Action: The Longevity Pathways
NAD+ is the obligate substrate for three major classes of enzymes involved in the Hallmarks of Aging.
¶ 1. Sirtuins: The "Guardians of the Genome"
Sirtuins (SIRT1–SIRT7) are a family of deacylases that require NAD+ to function. They cannot operate without it.
- SIRT1: Regulates circadian rhythms, mitochondrial biogenesis (via PGC-1α), and silences "junk" genes.[2]
- SIRT3: Localized in the mitochondria, it maintains antioxidant defenses (SOD2) and ensures efficient ATP production.
- SIRT6: Critical for DNA double-strand break repair and maintaining telomere stability.
- The Aging Link: Sirtuins have a low affinity for NAD+ (high ), meaning they are the first to shut down when levels drop. Restoring NAD+ "wakes up" these dormant defense systems.
¶ 2. PARPs: The DNA Repair Squad
Poly-ADP-ribose polymerases (PARPs), particularly PARP1, are the "first responders" to DNA damage.
- Mechanism: When PARP1 detects a strand break in DNA, it binds to the damage and uses NAD+ molecules to build long chains (poly-ADP-ribose) that signal other repair enzymes to fix the break.
- The Cost: Repairing a single DNA break can consume substantial amounts of NAD+.
¶ 3. CD38: The Immune Regulator
CD38 is a membrane-bound enzyme found on immune cells (macrophages, B cells) and widely in tissues during aging.
- Mechanism: It degrades NAD+ to produce cyclic ADP-ribose (cADPR), a molecule used for calcium signaling.
- The Problem: CD38 is an extremely inefficient enzyme—it burns roughly 100 molecules of NAD+ for every 1 molecule of cADPR signal it generates, acting as a massive "sink."[3]
¶ The Aging Problem: Why NAD+ Declines
The age-related collapse of NAD+ is not a simple production issue; it is a "supply and demand" crisis caused by two converging forces: increased consumption and decreased production.
¶ 1. The "Drain": Chronic Inflammation & CD38
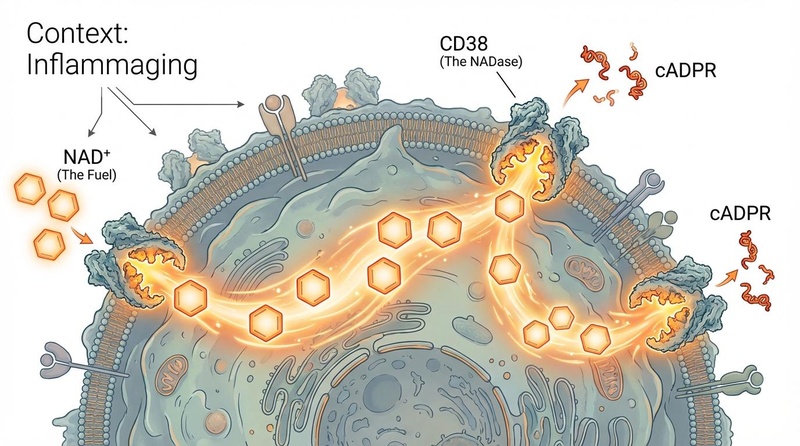
The most significant driver of NAD+ decline in older tissues is the upregulation of CD38.
- Inflammaging: As we age, accumulating senescent cells secrete inflammatory factors (SASP), such as TNF-α and IL-6.
- The Viscous Cycle: These inflammatory cytokines trigger macrophages (immune cells) to proliferate and express massive amounts of CD38. This creates a systemic "NAD+ sink," draining NAD+ not just from immune cells but from surrounding healthy tissues.[4]
- Evidence: In aged mice, CD38 levels in adipose and liver tissue can be 2–3x higher than in young mice.[5]
¶ 2. The "Burn": DNA Damage & PARPs
Aging is accompanied by an accumulation of DNA damage from UV radiation, oxidative stress, and replication errors.
- Chronic Activation: This constant low-level damage keeps PARP enzymes chronically active. They continuously draw from the NAD+ pool to fuel repairs.
- Hyperactivation: In conditions of severe damage, PARP1 can deplete cellular NAD+ to near-zero levels, causing energy failure and cell death (necroptosis).[6]
¶ 3. The "Bottleneck": NAMPT Decline
While consumption skyrockets, the body's ability to recycle NAD+ falters.
- NAMPT: The rate-limiting enzyme in the salvage pathway (see below) declines with age and metabolic stress.[7]
- Circadian Disruption: NAMPT expression is regulated by the circadian clock (BMAL1/CLOCK). Age-related sleep fragmentation and circadian misalignment suppress NAMPT, further choking off the supply.[8]
¶ Biosynthesis: How the Body Makes NAD+
Mammals rely on three pathways to maintain the NAD+ pool. In aging, the shift moves from efficient recycling to a desperate need for precursors.
¶ 1. The Salvage Pathway (Dominant)
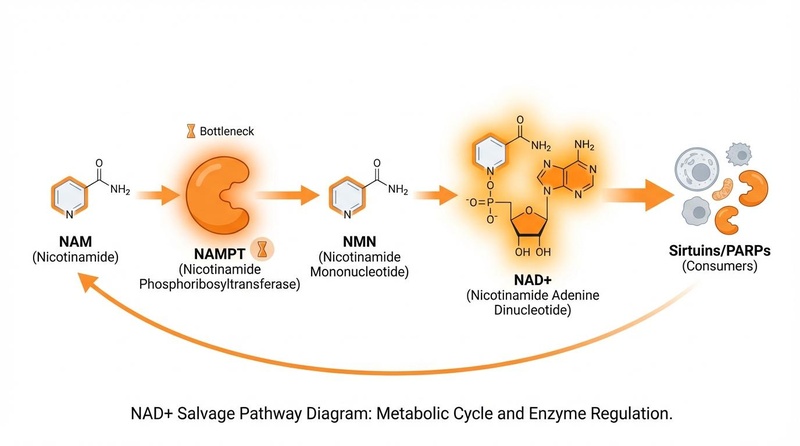
This pathway recycles the "waste" product of NAD+ usage—Nicotinamide (NAM)—back into fresh NAD+.
- Pathway: NAM → NMN (via NAMPT) → NAD+.
- Role: It provides the vast majority of NAD+ in most tissues.
- Vulnerability: Since it relies on NAMPT, this pathway is the most compromised by aging and inflammation.
¶ 2. The Preiss-Handler Pathway
This pathway utilizes dietary Nicotinic Acid (Niacin/Vitamin B3).
- Pathway: Nicotinic Acid (NA) → NaMN → NaAD → NAD+.
- Role: It bypasses the NAMPT bottleneck but is limited by the "flush" side effect of high-dose niacin and tissue-specific expression of its enzymes.[9]
¶ 3. The De Novo Pathway (Kynurenine)
This pathway builds NAD+ from scratch using the amino acid Tryptophan.
- Pathway: Tryptophan → ... (multi-step) ... → NAD+.
- Efficiency: Extremely low. It takes ~60 mg of Tryptophan to create 1 mg of Niacin equivalent. It is energetically expensive and cannot support the high demands of an aging body.[10]
¶ Summary of Decline Mechanisms
| Mechanism | Cause | Effect on NAD+ |
|---|---|---|
| CD38 Upregulation | Chronic inflammation (SASP) | Massive Consumption (Major Sink) |
| PARP Activation | Accumulated DNA damage | High Consumption (Variable Sink) |
| NAMPT Downregulation | Aging, circadian disruption | Reduced Recycling (Supply Failure) |
Clinical Implication: Effective restoration of NAD+ likely requires addressing both sides of the equation: supplying precursors (to bypass NAMPT) and reducing inflammation (to lower CD38).
👉 For actionable strategies to restore NAD+, see NAD+ Precursors.
¶ References
Covarrubias, A. J., Perrone, R., Grozio, A., & Verdin, E. (2021). NAD+ metabolism and its roles in cellular processes during ageing. Nature Reviews Molecular Cell Biology, 22(2), 119-141. https://www.nature.com/articles/s41580-020-00313-x ↩︎
Imai, S., & Guarente, L. (2014). NAD+ and sirtuins in aging and disease. Trends in Cell Biology, 24(8), 464-471. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4112140/ ↩︎
Chini, E. N. (2009). CD38 as a regulator of cellular NAD: a novel potential pharmacological target for metabolic conditions. Current Pharmaceutical Design, 15(1), 57-63. https://pubmed.ncbi.nlm.nih.gov/19149601/ ↩︎
Chini, C. C., et al. (2020). CD38 ecto-enzyme in immune cells is induced during aging and regulates NAD+ and NMN levels. Nature Metabolism, 2, 1284–1304. https://www.nature.com/articles/s42255-020-00298-z ↩︎
Camacho-Pereira, J., et al. (2016). CD38 Dictates Age-Related NAD Decline and Mitochondrial Dysfunction through an SIRT3-Dependent Mechanism. Cell Metabolism, 23(6), 1127-1139. https://www.cell.com/cell-metabolism/fulltext/S1550-4131(16)30224-8 ↩︎
Pacher, P., & Szabo, C. (2008). Role of the peroxynitrite-poly(ADP-ribose) polymerase pathway in human disease. American Journal of Pathology, 173(1), 2-13. https://pmc.ncbi.nlm.nih.gov/articles/PMC2438291/ ↩︎
Yoshino, J., Mills, K. F., Yoon, M. J., & Imai, S. (2011). Nicotinamide mononucleotide, a key NAD+ intermediate, treats the pathophysiology of diet- and age-induced diabetes in mice. Cell Metabolism, 14(4), 528-536. https://www.cell.com/cell-metabolism/fulltext/S1550-4131(11)00345-8 ↩︎
Ramsey, K. M., et al. (2009). Circadian clock feedback cycle through NAMPT-mediated NAD+ biosynthesis. Science, 324(5927), 651-654. https://pubmed.ncbi.nlm.nih.gov/19342540/ ↩︎
Bogan, K. L., & Brenner, C. (2008). Nicotinic acid, nicotinamide, and nicotinamide riboside: a molecular evaluation of NAD+ precursor vitamins in human nutrition. Annual Review of Nutrition, 28, 115-130. https://pubmed.ncbi.nlm.nih.gov/18429699/ ↩︎
Badawy, A. A. (2017). Kynurenine Pathway of Tryptophan Metabolism: Regulatory and Functional Aspects. International Journal of Tryptophan Research, 10. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5398323/ ↩︎